Axonics® Announces Positive Top-Line Clinical Data from its ARTISAN-SNM Pivotal Study
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20190219005354/en/
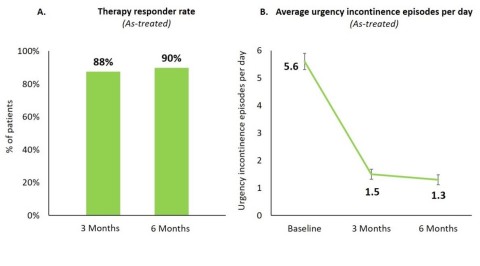
Figure 1A & 1B
The clinical study demonstrated that patients implanted with the Axonics r-SNM System received clinically meaningful and statistically significant improvements in Urinary Urgency Incontinence (“UUI”) symptoms and quality of life. Additionally, the study met all secondary endpoints. No serious device-related adverse events have been reported.
The ARTISAN-SNM study is a 129-patient single-arm, prospective,
multi-center, unblinded pivotal clinical study approved under an
Top-Line Results
All patients diagnosed with UUI and meeting study criteria were implanted with a tined lead and the Axonics neurostimulator. Efficacy data was collected using a 3-day bladder diary, a validated quality of life questionnaire (ICIQ-OABqol), and a satisfaction questionnaire. Therapy responders were identified as patients with at least 50% reduction in urgency incontinence episodes at follow up visits as compared to baseline. An as-treated analysis was performed for all 129 implanted patients.
At six months, 90% of all implanted patients were therapy responders (Figure 1A) including 80% of therapy responders with a ≥75% reduction in urgency incontinence episodes of which 34% were completely dry.
Across all patients, urgency incontinence episodes per day reduced from 5.6 ± 0.3 (mean ± standard error) at baseline to 1.3 ± 0.2 at six months (p<0.0001; Figure 1B).
Patients averaged statistically and clinically significant improvement on the composite ICIQ-OABqol score (34 points) at six months as compared to baseline and 93% of all implanted patients were satisfied with their r-SNM therapy.
Premarket Approval (PMA) Status with the U.S.
Axonics filed a PMA application on
Conference Call and Webcast
As previously announced, the Company will host a conference call with
the investment community to discuss 2018 fourth quarter and full-year
financial results and recent business developments, including clinical
data from the ARTISAN-SNM study, on
Interested parties may access the live call via telephone by dialing (866) 687-5771 (U.S.) or (409) 217-8725 (International) and using passcode 3386378. A live webcast of the call may be accessed by visiting the Events & Presentations page of the investors section of the Company's website at ir.axonicsmodulation.com. A replay of the webcast will be available shortly after the conclusion of the call and will be archived on the Company's website for 90 days.
About Overactive Bladder and Sacral Neuromodulation
Overactive bladder (OAB) includes urinary urge incontinence and urinary
frequency and affects an estimated 85 million adults in the U.S. and
About
Axonics, based in
Forward-Looking Statements
Statements made in this press release that relate to future plans,
events, prospects or performance are forward-looking statements as
defined under the Private Securities Litigation Reform Act of 1995.
Words such as “planned,” “expects,” “believes,” “anticipates,”
“designed,” and similar words are intended to identify forward-looking
statements. While these forward-looking statements are based on the
current expectations and beliefs of management, such forward-looking
statements are subject to a number of risks, uncertainties, assumptions
and other factors that could cause actual results to differ materially
from the expectations expressed in this press release, including the
risks and uncertainties disclosed in Axonics filings with the
______________________
1 The Axonics r-SNM System is
currently designated as an investigational medical device
View source version on businesswire.com: https://www.businesswire.com/news/home/20190219005354/en/
Source:
Axonics Modulation Technologies, Inc.
Dan Dearen, +1-949-396-6320
President
& Chief Financial Officer
ir@axonics.com
Investor & Media Contact
W2Opure
Matt Clawson,
+1-949-370-8500
mclawson@w2ogroup.com
